Cyanide is a rapidly acting, deadly chemical that is highly toxic to chickens. Cyanide poisoning can occur through exposure to fumes from fires, or ingestion of certain common foods, plants, or products containing cyanide or cyanogenic glycosides. Cyanide prevents the cells of the body from using oxygen. When this happens, the cells die. Cyanide acts almost instantly, and can cause death within a few seconds to minutes.
Cyanide combines with iron in cytochrome oxidase, preventing terminal electron transfer and blocking cellular respiration. Oxyhemoglobin cannot release oxygen. Although the blood is highly oxygenated and therefore a characteristic bright cherry red color, oxygen delivery to the tissues is poor and the bird becomes severely hypoxic.
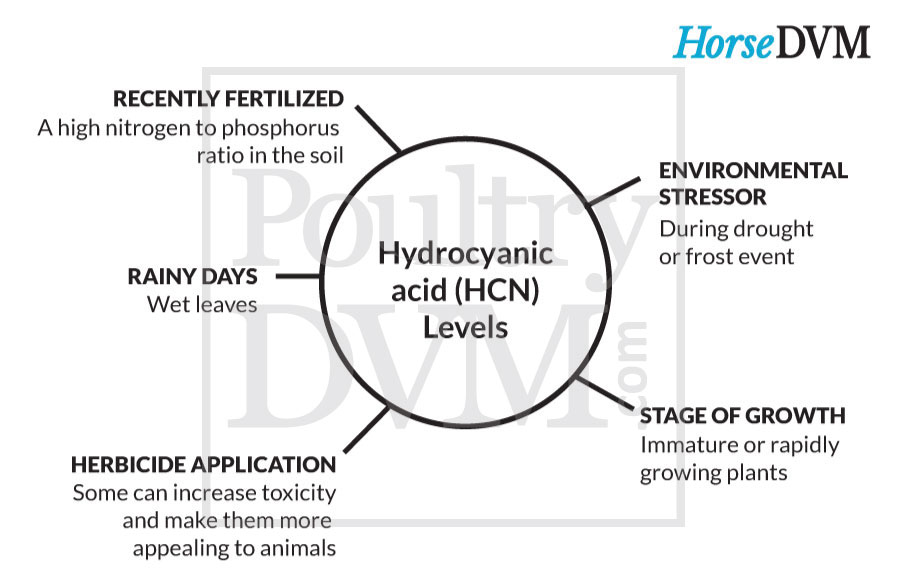
A number of different plants can accumulate cyanogenic glycosides. The amount of cyanide present in these plants are highly variable, but are typically highest during the spring while blooming. Drought and dry growing conditions enhance the accumulation of cyanogenic glycosides in many of these plants as well.
Plants and Foods which Contain Cyanogenic Glycosides
Plants capable of producing high levels of cyanide include:
Signs of Cyanide Poisoning
Chickens that die from cyanide poisoning have bright red, oxygenated blood and bodily tissues are usually congested with blood. The reported oral LD50 for domestic chickens is 11.1 mg/kg of body weight. Chickens will usually die within 15 to 30 minutes of ingestion of the toxic substance.
Treatment for Cyanide Poisoning in Chickens
Cyanide poisoning is treated with specific antidotes and supportive medical care. To be effective, an antidote must be administered before there is irreversible cell damage, often within minutes of exposure.
Treatment involves administering intravenous amyl and/or sodium nitrate. Those substances combine with hemoglobin to produce methemoglobin, which, in turn, has a strong affinity for cyanide. Sodium thiosulfate is then administered to scavenge the cyanide from the methemoglobin, producing thiocyanate. Hydroxocobalamin (Vitamin B12a) can also be used, which acts by combining with unbound cyanide to form cyancobalamin.